Appendix 2: Calculation of blood and plasma dilution
The following algorithm is used for determining the suitability of post-transfusion/infusion samples for mandatory screening for transmissible infections. It is based on the algorithm developed by the Food and Drug Administration (FDA).
Step 1: Determine which sample is suitable for testing
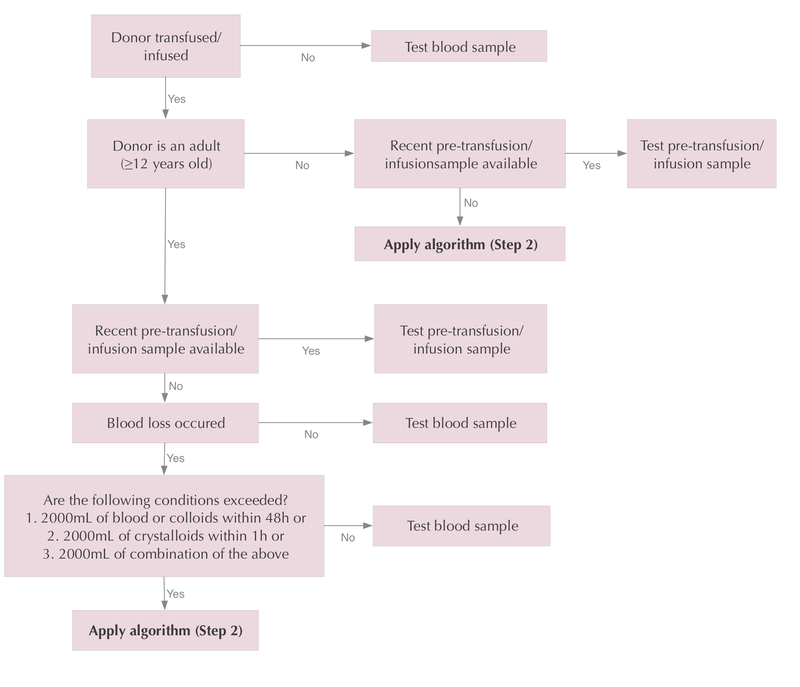
Figure 1: Decision process
Use this flow chart to determine whether a blood sample or a pre-transfusion/infusion sample should be tested, or whether the algorithm (Step 2) should be applied.
Step 2: Algorithm to assess dilution
Step 2a: Calculate volumes
1. Calculate donor plasma volume
Donor weight (kg) ÷ 0.025 = donor plasma volume (mL)
2. Calculate donor blood volume
Donor weight (kg) ÷ 0.015 = donor blood volume (mL)
3. Record volume of blood transfused
(in the 48 hours prior to death or sample collection, whichever comes first)
Add the following to calculate the volume of blood transfused (mL):
- Red blood cells in 48 hours (mL)
- Whole blood in 48 hours (mL)
- Reconstituted blood in 48 hours (mL)
The volume of blood transfused (mL) is Sum A.
4. Record volume of colloid infused
(in the 48 hours prior to death or sample collection, whichever comes first)
Add the following to calculate the volume of colloids transfused (mL):
- Plasma in 48 hours (mL)
- Platelets in 48 hours (mL)
- Albumin in 48 hours (mL)
- Hydroxyethyl starch (HES) or other colloids in 48 hours (mL)
The volume of colloids transfused (mL) is Sum B.
5. Record volume of crystalloid infused
(in the 1 hour prior to death or sample collection, whichever comes first)
The volume of crystalloid infused (mL) is Sum C.
Step 2b: Assess dilution
1. Calculate volume of plasma transfused
Sum B + Sum C = volume of plasma transfused (mL)
2. Calculate total volume transfused
Sum A + Sum B + Sum C = total volume transfused (mL)
3. Calculate plasma dilution
Is the volume of plasma transfused greater than the donor plasma volume?
- Yes
- No
4. Calculate blood dilution
Is the total volume transfused greater than donor blood volume?
- Yes
- No
5. Determine sample suitability
If the answers to both questions above are 'No', the post-transfusion/infusion sample is acceptable.
If the answer to either question above is 'Yes', use a pre-transfusion/infusion sample. If a suitable sample is not available, seek expert advice and inform transplant centre, testing laboratory, tissue bank as necessary.
Last updated in TD-DSG Edition 203 Release 32 (26 July 2018)